-
Passive Nanogold Particles: Still the Gold Standard for Lateral Flow Assays
In lateral flow assay (LFA) development, new label technologies emerge every year. While it’s tempting to assume they offer an edge, in reality, most diagnostics teams racing to prototype, validate, and commercialize an assay don’t need more complexity. They need consistency, scalability, and a material that works reliably across many assay formats and manufacturing environments. (more…)
-

Move Faster and Smarter: Why Working with a Contract Developer Can Accelerate Your Go-To-Market Strategy
In the fast-evolving world of diagnostics and biotechnologies, time-to-market is everything. For companies of all sizes—whether resource-rich or resource-limited—contract assay developers offer a path to faster timelines, reduced risk, and expert guidance. (more…)
-
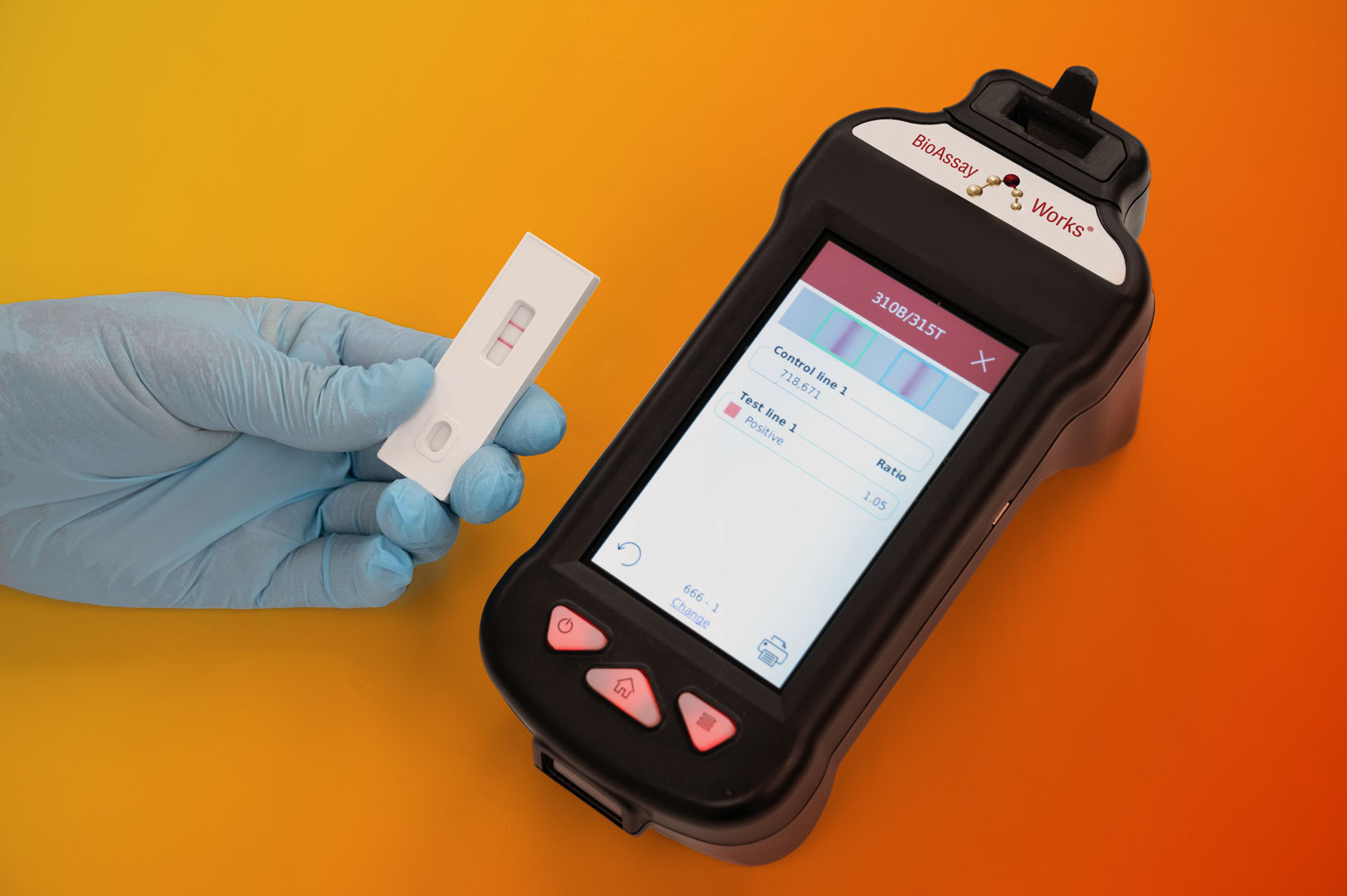
Are Lateral Flow Assays Ready for the Leap from Qualitative to Quantitative? What You Need to Know.
As demand for more precise healthcare diagnostics grows, the landscape is shifting. Today, in certain contexts, there’s a strong push for LFAs to move beyond simple “yes or no” answers and deliver accurate, quantitative results that can support critical clinical decisions. (more…)
-

Securing Supply Chains for Rapid Diagnostics During Global Disruption
The current U.S.-China trade war has reignited global concerns over the stability and security of supply chains — especially for critical sectors like diagnostics. For companies developing lateral flow assays and point-of-need tests, even small interruptions in material sourcing can result in delayed launches, failed partnerships, and lost revenue. (more…)