Are Lateral Flow Assays Ready for the Leap from Qualitative to Quantitative? What You Need to Know.
Lateral flow assays (LFAs) have long been the go-to solution for point-of-care testing. Their appeal is clear: LFAs are affordable, easy to use, and robust enough to deliver rapid, reliable screening results. However, as the demand for more precise healthcare diagnostics grows, the landscape is shifting. Today, in certain contexts, there’s a strong push for LFAs to move beyond simple “yes or no” answers and deliver accurate, quantitative results that can support critical clinical decisions. So much so that quantitative LFAs represent a growing segment of the $11 billion LFA diagnostic market.
What’s driving this evolution, and what challenges must be overcome to turn LFAs into true quantitative tools? Here’s what you need to know.
From Qualitative to Quantitative: What’s the Difference?
A qualitative assay simply tells you whether a target analyte is present in a sample. In contrast, a quantitative assay provides a numerical measurement—an exact amount or concentration of the analyte. To achieve this, the test must generate a detectable signal that increases (in sandwich assays) or decreases (in competitive assays) in direct proportion to the analyte’s concentration. This relationship should be linear or at least reliably calibrated over a defined range. Establishing a calibration curve using known standards is essential, as it allows users to determine the concentration in unknown samples.
The Challenge for LFAs
Traditional LFAs, while excellent for quick screening, haven’t historically met the rigorous requirements for quantitative testing. Unlike ELISA tests, which run calibrated standards alongside unknown samples, LFAs typically process one sample at a time without internal controls. This raises the question: how can LFAs deliver accurate, quantitative results without sacrificing their core advantages?

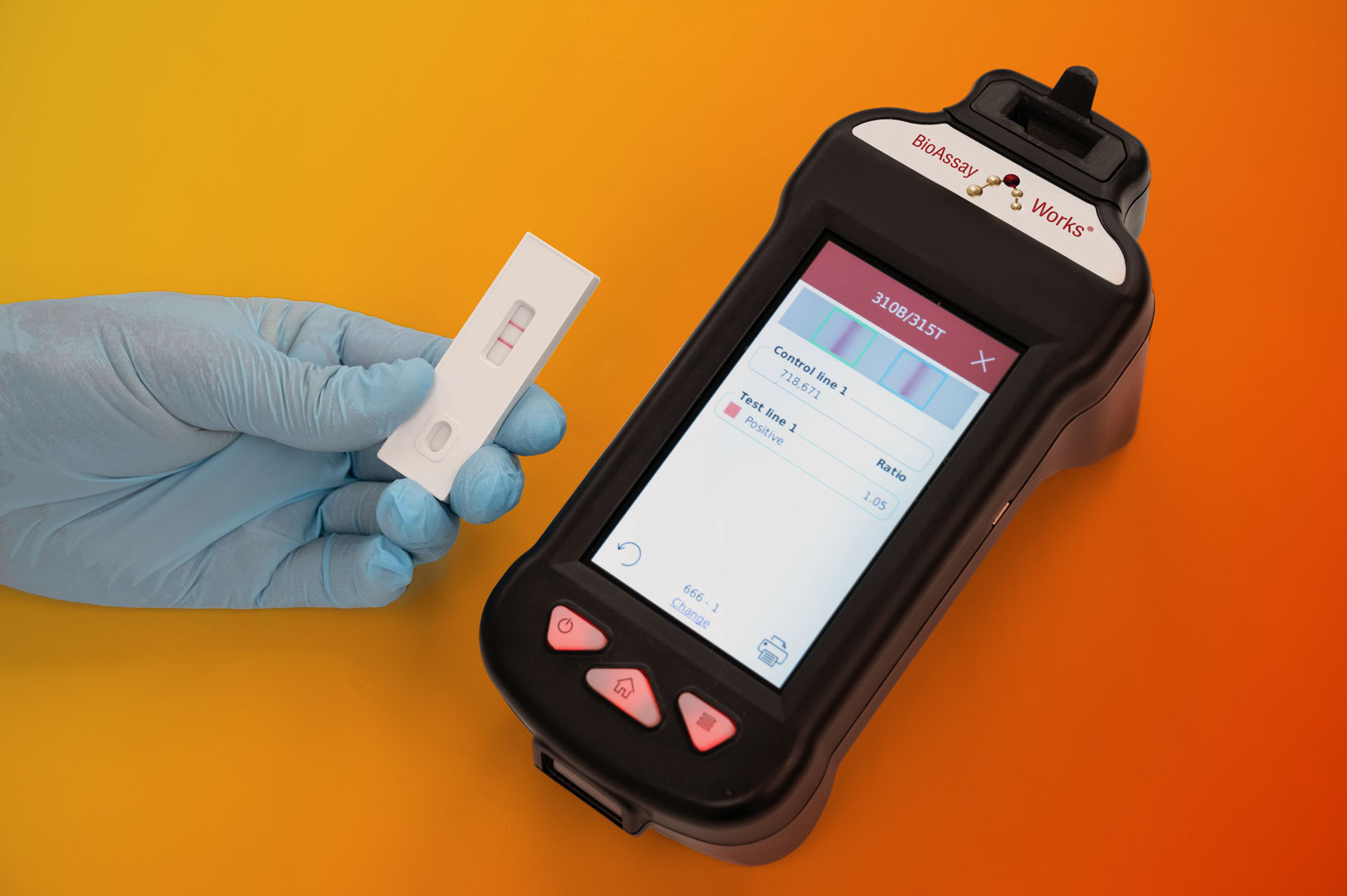
Upgrading LFAs: The Role of Reader Devices
The most common approach to making LFAs quantitative is to pair them with an external reader — either a dedicated laboratory instrument or, more recently, a smartphone app. These devices measure the intensity of the test signal and use algorithms to translate that signal into a precise value. For the system to be reliable, both the test and the reader must be validated together to ensure clinical accuracy, sensitivity, specificity, and linearity across the measurement range.
What Makes a Truly Quantitative LFA?
A lateral flow system can be considered genuinely quantitative only if the combination of the assay and the reader provides accurate, consistent, and clinically meaningful results. The system’s limitations will always hinge on the assay’s ability to generate a readable signal. While reader devices reduce user-to-user variability, they also introduce their own challenges, such as sensitivity to ambient light or differences between phone cameras, in the case of apps.
The Trade-Off: Simplicity vs. Precision
Choosing to use a quantitative over a qualitative test depends on the need or circumstance. Moving to a quantitative platform often means giving up some of the traditional benefits of qualitative LFAs — like lower cost, simplicity, and not needing additional equipment or software — in exchange for greater precision and clinical utility.

Real-World Impact
When field testing demands quantitative results:
VMRD developed a test to measure Serum Amyloid A (SAA) in equine, canine, and feline blood. Given the particular challenges of veterinary medicine, having a truly patient-side test with quantitative results provides veterinarians with rapid, actionable information that can make an immediate impact on their clinical assessments and treatment decisions.
- Early identification of infection is challenging, and initial signs are often subtle, therefore sensitive and accessible tools are needed.
- SAA levels in blood are rapidly responsive to clinical changes, increasing dramatically within 6-12 hours with acute, systemic inflammation.
- Tracking the quantitative SAA value over time with the VMRD LFA enables rapid and objective monitoring of clinical condition and treatment effectiveness.
- A user-friendly handheld reader allows veterinarians to easily run the test wherever their patients may be, including vet clinics, barns, and even outdoors.
The Bottom Line:
As healthcare continues to demand faster and more accurate diagnostics, the growth of quantitative testing is inevitable. The key is finding the right balance between maintaining LFA’s traditional simplicity while embracing the need for precise, actionable results. The future of point-of-care testing may well depend on it.